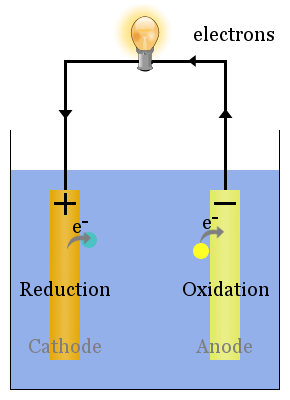
The anode is negative in an electrochemical cell due to the negative potential with respect to the solution. The anode is positive and cathode negative in an electrolyte cell. The anode is negative and the cathode is positive in a galvanic or voltaic cell. The positive is the cathode and the negative is anode during the discharge. The positive is the anode and the negative a cathode during the charge. Cathode attracts cations or positive charges. Cathode becomes an anode in a galvanic cell. Anode becomes cathode in a galvanic cell. 
Reduction takes place at the cathode in an electrolyte cell. Oxidation occurs at the anode in an electrolyte cell.The cathode is the terminal where electricity leaves the device. The anode is the terminal where electricity enters the device.Main Differences between Anode and Cathode The anode is a positive terminal in an electrolyte cell. The anode is a negative terminal in a galvanic cell and electrons move towards the external part of the circuit. The negative anions usually react to give off electrons. But anode is defined as a positively charged terminal where current flows into the device.Īn anode in electrochemistry is a terminal where oxidation or loss of electrons occurs. It is a type of electrode that is either positive or negative depending on the type of cell. Reduction reaction occurs at the cathode.īecome a negative charge in a galvanic or voltaic cells.īecome positive charge in a galvanic or voltaic cell.Īttracts positive cations and rejects negatively charged anions. It is an electrode that carries electrical current from the negative terminal to the positive terminal. It is an electrode that carries conventional current from the positive terminal to the negative terminal. You May Also Like: Difference between Acid and Base Comparison Table (Anode vs Cathode) Basic Terms To clear out the confusion, this article provides further differences between anode and cathode. However, these components can be interchanged during a reversible process. So, what is the main difference between anode and cathode? The former is a terminal where the conventional current flows into a device from outside while the latter is the terminal where the conventional current flows out of the device. These components help to describe the flow of current within a circuit. The main components of an electrode are cathode and anode. The substance provides a medium where electric currents enter and leave the non-metallic parts like an electrolyte cell. An electrode is a substance that helps to conduct electricity by establishing electrical contact with non-metallic parts of the circuit.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
